Neurodiversity in Action Understanding the brain basis of dyslexia
Rebecca A. Marks, Ph.D.
Assistant Professor, Purdue University
July 2025
Dyslexia, a specific learning disability (SLD) characterized by word reading impairment, is commonly misunderstood as a disorder in which learners simply reverse their letters, like b and d. Research in cognitive neuroscience tells a different story, and can help us to target our instruction to better serve struggling readers.
The reading brain
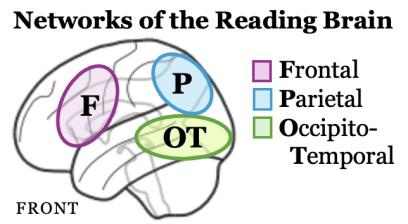
Reading involves integrating brain systems for language with brain systems for visual perception. A 5-year-old child comes to the task of learning to read with years of language experience; learning to read involves connecting their knowledge of the sounds and meanings of language to printed symbols on a page.
The reading brain can be broken down into three main systems that work together in a larger network. The frontal circuit (purple) analyzes the sounds within words; the parietal circuit (blue) connects sound, meaning, and print; and the occipito-temporal circuit (green) supports fluent, automatic visual word recognition. Early reading relies heavily on the frontal regions of the brain to support effortful phonological decoding. Skilled readers still use this frontal network, but rely more heavily on regions in the back of the brain, reflecting efficient connections between word meaning and printed form.
Defining dyslexia
Dyslexia is characterized by persistent difficulties in accurate and/or fluent word reading despite adequate instruction and intelligence. There is no single root cause of dyslexia, and children with dyslexia have a wide range of cognitive profiles. One frequent struggle is in phonological processing, or the ability to accurately perceive and manipulate the sounds of language. Many children also struggle with rapid automatized naming, or RAN, which assesses the speed of naming familiar items like letters or numbers. Dyslexia also has high heritability, which means that it has a genetic component and runs in families (Doust et al., 2022).
The cognitive neuroscience of dyslexia
Research in cognitive neuroscience has revealed differences in brain responses to spoken language as well as written language (D’Mello & Gabrieli, 2018). On average, individuals with dyslexia under-activate the parietal (blue) and occipito-temporal (green) regions at the back of the brain that support connecting letters and sounds to print. These brain differences are present long before children learn to read. Studies have found that infants who are diagnosed with dyslexia many years later show neurocognitive differences in their responses to speech sounds as infants (Leppänen et al., 2002; Yu et al., 2018).
It is important to understand that these brain differences in dyslexia are based on group averages. Our most advanced science to date cannot determine whether or not an individual has a reading disorder based on a brain scan alone. This is because there is so much variability in the patterns of brain activity within groups of readers, both skilled and impaired.
Consider this analogy: A class of second graders will be shorter on average than a class of third graders. Even so, if all you know about a child is their height, you couldn’t reliably place them in the correct grade. Likewise, an individual brain image is insufficient to diagnose dyslexia.
Pictured below are brain activations in the left hemispheres of six children (grades 3–5) during fMRI neuroimaging. Each child completed a task that involved reading two words and deciding whether they rhyme (e.g., ocean | motion). The images below show the regions of the brain that were more active during this reading task than during a non-reading task.
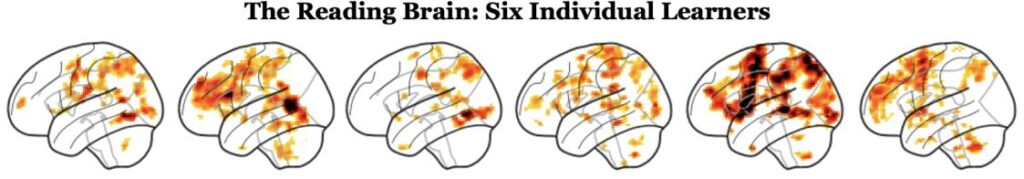
Of the six brains above, half belong to children who are reading at grade level. The other half are struggling readers with dyslexia. We can’t tell from a brain image alone whether a child has dyslexia.
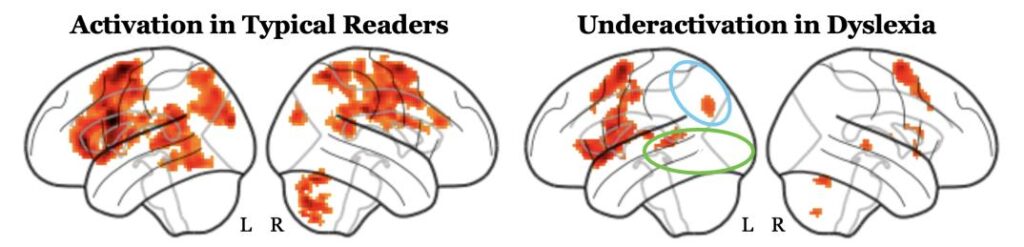
When combined as a group, general patterns of under-activation in dyslexia become clearer. Below is the result of averaging brain activations during word reading from 50 typical readers on the left, and 63 readers with dyslexia on the right. We can see reduced engagement on average in the parietal (blue) and occipito-temporal (green) regions.
How can neuroscience inform practice?
Cognitive neuroscience is one piece of a much larger puzzle informing approaches to education. Neuroscience tells us about the mechanisms underlying reading development and learning differences. When we understand these mechanisms, we can target instruction to support them.

Many dyslexic individuals describe experiences of “word blindness,” or feeling as if words and letters swim in front of their eyes as they try to read. The idea that dyslexia is, at its core, related to oral language rather than vision may feel counter-intuitive. Neuroscience revealed, for the first time, that dyslexia was associated with differences in auditory phonological processing, even in the absence of print (Kovelman et al., 2012).
Building on this knowledge, we can prioritize reading instruction that best supports the brain mechanisms that differ in dyslexia. Children who go on to develop dyslexia show reduced brain responses during phonological processing long before they learn to read (Leppänen et al., 2002; Yu et al., 2018). Armed with this understanding, we can provide systematic, explicit instruction that supports phonological decoding. Optimal early reading instruction builds automaticity in word recognition, allowing children to reallocate brain resources away from effortful decoding and towards meaning-making.
Best practices in reading instruction
The cognitive neuroscience of reading provides little direct insight into classroom practice. However, following the guiding principles below may help teachers and clinicians to best support their struggling learners. Ideally, instruction should be:
like the gears of a machine.
Explicit, clear, and direct:
- De-mystifies relationships between sounds and letters
- Breaks words down into their meaningful parts
- Teaches oral language skills and relates them to reading, e.g., comparing spoken sentences to the complex syntax often found in academic texts
- Models skills and provides opportunities for practice
Sequenced and cumulative:
- Follows a logical scope and sequence that allows students to build on their learning over time
- Builds on students’ growing vocabulary and background knowledge
Data driven:
- Informed by ongoing low-stakes assessment
- Uses student data to track learning progress and target areas of challenge
Further Reading
To learn more about the brain basis of dyslexia:
Kearns, D. M., Hancock, R., Hoeft, F., Pugh, K. R., & Frost, S. J. (2019). The neurobiology of dyslexia. Teaching Exceptional Children, 51(3), 175-188.
To learn more about Rapid Automatized Naming (RAN) and its role in dyslexia screening:
What Educators Need To Know About RAN (Norton, 2020)
For an overview of explicit instruction in literacy:
Vaughn, S. & Fletcher, J. (2021). Explicit instruction as the essential tool for executive the science of reading. The Reading League Journal, 2(2), 4-11.
References
D’Mello, A. M., & Gabrieli, J. D. (2018). Cognitive neuroscience of dyslexia. Language, speech, and hearing services in schools, 49(4), 798-809.
Doust, C., Fontanillas, P., Eising, E., Gordon, S. D., Wang, Z., Alagöz, G., … & Luciano, M. (2022). Discovery of 42 genome-wide significant loci associated with dyslexia. Nature genetics, 54(11), 1621-1629.
Kovelman, I., Norton, E. S., Christodoulou, J. A., Gaab, N., Lieberman, D. A., Triantafyllou, C., … & Gabrieli, J. D. (2012). Brain basis of phonological awareness for spoken language in children and its disruption in dyslexia. Cerebral Cortex, 22(4), 754-764.
Leppänen, P. H., Richardson, U., Pihko, E., Eklund, K. M., Guttorm, T. K., Aro, M., & Lyytinen, H. (2002). Brain responses to changes in speech sound durations differ between infants with and without familial risk for dyslexia. Developmental neuropsychology, 22(1), 407-422
Yu, X., Raney, T., Perdue, M. V., Zuk, J., Ozernov‐Palchik, O., Becker, B. L., … & Gaab, N. (2018). Emergence of the neural network underlying phonological processing from the prereading to the emergent reading stage: A longitudinal study. Human brain mapping, 39(5), 2047-2063.